What is pH level?
pH is an important property of a solution that indicates if the solution is acidic or basic in nature. The pH of the solution determines the availability of nutrients, biological functions, and microbial activity and impacts the behaviour of chemicals. Furthermore, it helps to identify the intensity of solution and categorize them into Acidic, Basic and Neutral.
pH Value is used to measure the concentration of hydrogen ions (H+) in water. The ‘H’ in pH is Hydrogen and ‘p’ refers to Power. Naturally, pH is said to indicate the Power of Hydrogen in a given sample solution.
pH is an important and essential quantitative measure, that reflects the chemical nature of a solution. The pH can regulate the levels of nutrients, microbial activity, biological functions, and the behaviour of the chemicals.
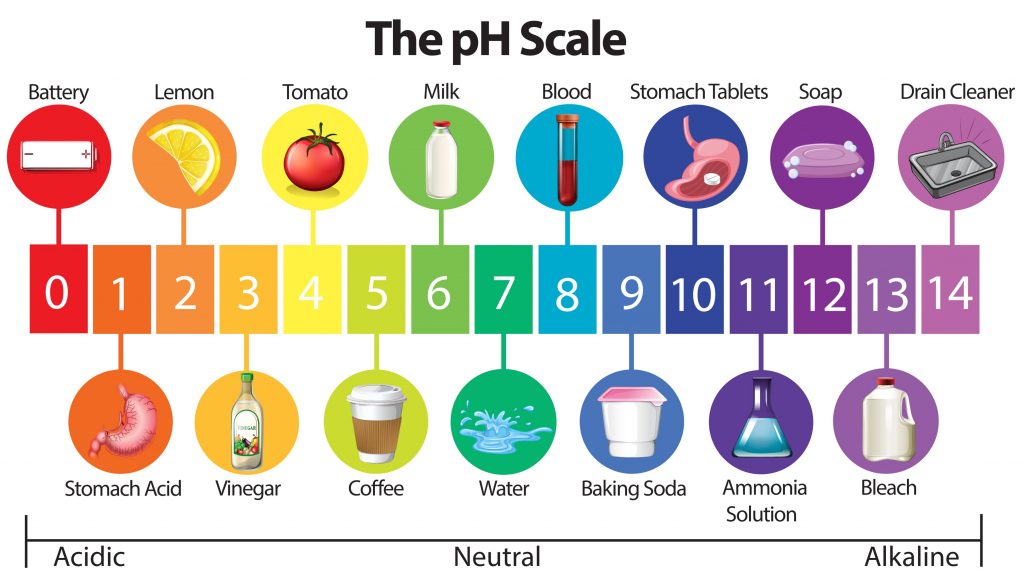
pH scale – The Acids, The Bases, and The Neutral
The pH scale is a 14 pointer scale. Having a pH less than 7 will classify a solution as acidic. For example, lemon juice or citric acid has a pH of 2 making it a highly acidic solution. Similarly, having a pH over 7 will land a solution in the base category like bleach, with a pH of 12. There is however a balance or a neutral pH value, this suggests that the solution is neither acidic nor basic in its nature.
 |
How can one identify the pH levels of a solution?
The Litmus Test
- There are several ways in which one can identify the pH levels of a solution, the most common and easiest being the ‘Litmus Test’. The test essentially uses a strip of paper that is made from lichens. This strip of paper changes its color based on the characteristics of the solution it interacts with. Light blue litmus paper turns red under acidic conditions and red litmus paper turns blue under basic or alkaline conditions. A neutral litmus test would result in a purple shade.
- pH paper, however, comes with some restrictions. First, it is not an accurate indicator of pH and does not give us a numerical pH value. Instead, it roughly indicates the nature of the sample.
pH Meter
- Another way is the pH meter, which is a much more articulate and accurate method of testing. A pH meter is a scientific apparatus that can not only identify if a solution is acidic or alkaline but also give us an exact measure of its pH levels.
How is a pH test relevant and important?
- One of the common usages of these pH tests and indicators is acid-base titration. A process of quantifying the concentration of acid or base in a solution by neutralising it with the exact same proportion of base or acid.
- In situations like these where one wants to know the exact value, digital pH indicators have proven to be the preferred choice.
- The titration of a strong acid – strong base is a method used to determine the concentration of the acidic solution by titrating it with a basic solution of known concentration, or vice-versa. When a strong acid reacts with a strong base, they completely neutralise each other, this results in the residue of salt and water.
- A weak acid and strong base or vice-versa make the titration curve irregular and the pH shifts at a slower rate with small additions of titrant near the equivalence point.
Different industries, different uses
pH monitoring is essential in several industries, like wastewater treatment, aquaculture, pharmaceuticals, food and beverages, paper, and the textile industries. Each industry has a specific pH level prescribed for their products to remain safe for use.
-
Wastewater
 |
While treating wastewater, several acidic substances are removed from the water. The pH levels are adjusted with the use of chemicals and are monitored at every phase of the treatment so that the treated water is safe and fit to reuse once the process is complete.
-
Aquaculture
 |
In the aquaculture industry, it becomes crucial for the industry to maintain the right pH levels to ensure safe and consumable seafood. Therefore, in this case too, the pH level is monitored very closely.
-
Food and Beverage
 |
Just like the aquaculture industry, the food and beverage industry is also an industry that keeps a close check on the pH levels at different stages of manufacturing. Observing the pH levels of bottled water and meat products is a common and encouraged practice.
-
Paper and Textile
 |
Paper and textile industries produce large quantities of wastewater that can be harmful to their equipment and the environment. So, it’s their moral responsibility to keep an eye on the pH levels of this wastewater.
Keeping a close eye on the pH levels
All these industries need to very closely monitor and maintain their desired pH levels which makes it important to use an apparatus that is accurate and eliminates chances of human errors.
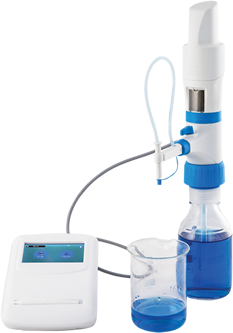
Microlit E-Burette helps deliver precise samples and guarantees accuracy to industrial titrations. This can be credited to its state-of-the-art design and precision. It exhibits excellent chemical compatibility and helps in performing precise titrations with reliability in practical laboratory environments.
To know more about the Microlit E-Burette, you can write to us at info-usa@microlit.com.




 25287
25287